Electrode Slurry Preparation, Rheological Control, and Evaluation in Lithium-Ion Battery Manufacturing
A detailed engineering article on lithium-ion battery electrode slurry preparation routes, rheological control, dispersion factors, evaluation methods, and SWCNT relevance.
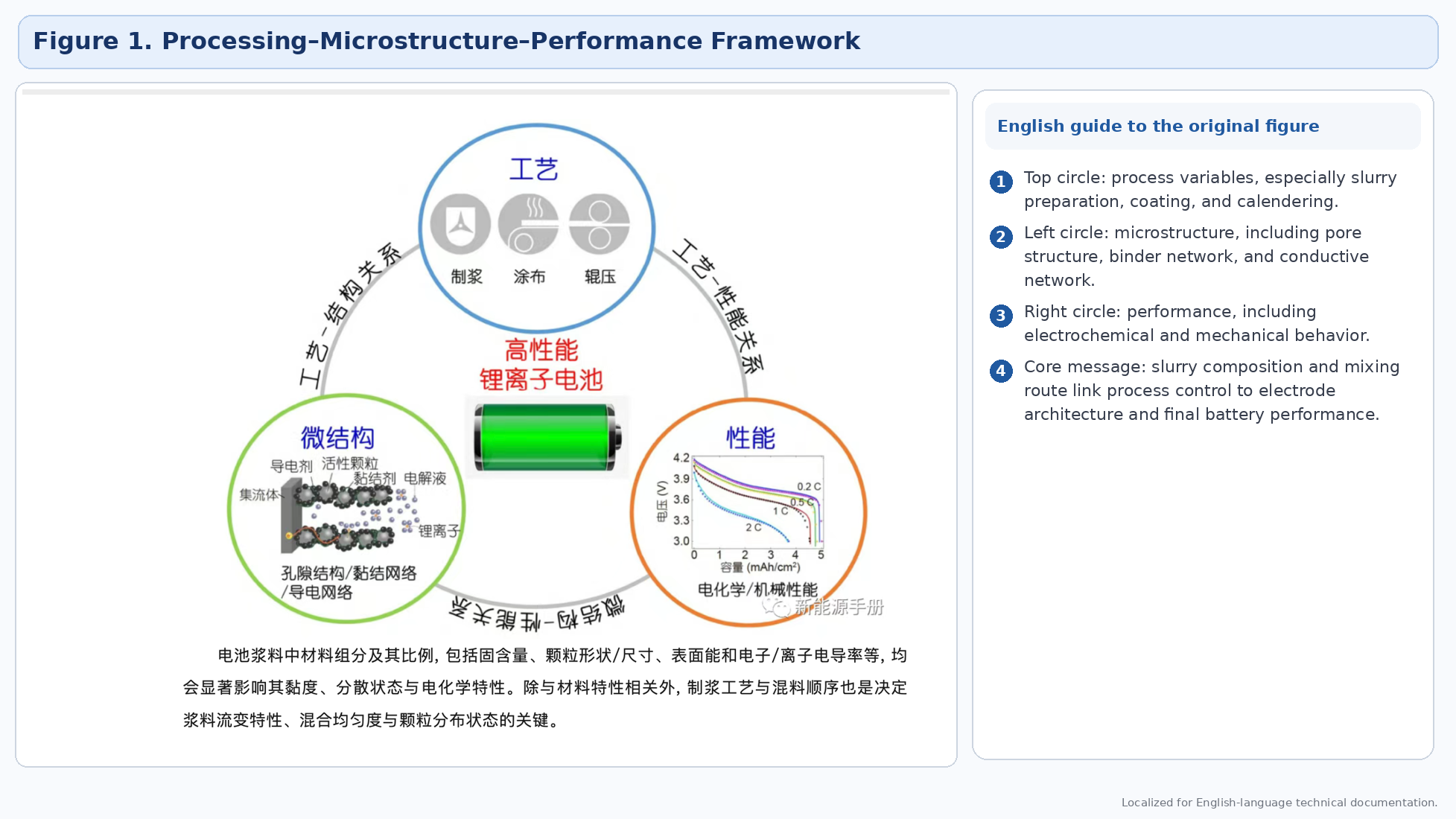
Note on figures. This revised edition replaces the earlier raw image insertions with localized English-ready figure panels. Where the original figure contains Chinese labels, the document now adds a clean English interpretation panel beside the source graphic so the figure remains usable for English-language readers.
1. Why slurry preparation matters
Electrode slurry is not only a transport medium for active material, conductive additive, and binder. It is also the first place where the internal structure of the future electrode begins to form. In practical battery manufacturing, the quality of slurry preparation directly influences coating stability, drying behavior, electrode microstructure, and the consistency of the finished cells.
The original article correctly frames slurry as a bridge between process conditions, microstructure, and battery performance. When the slurry formulation or mixing route is unstable, the result can be non-uniform conductive pathways, uneven binder distribution, local particle agglomeration, unstable adhesion, and poorer electrochemical performance.

2. Key challenges in electrode-slurry research
Lithium-ion battery slurry is a non-equilibrium suspension system. As energy-density requirements rise and formulations move toward smaller particle sizes and higher loading, the risk of agglomeration and internal non-uniformity increases. If active particles, conductive additives, and binders are not distributed uniformly in the wet slurry, the dried electrode can inherit that disorder as uneven local resistance, inferior rate performance, and reduced durability.
Because most battery slurries are opaque, their internal structure cannot be judged reliably by eye. Rheological analysis therefore becomes one of the most useful indirect windows into internal dispersion. The article repeatedly links slurry stability with raw-material feeding order, solvent type, solids content, and mixing conditions.
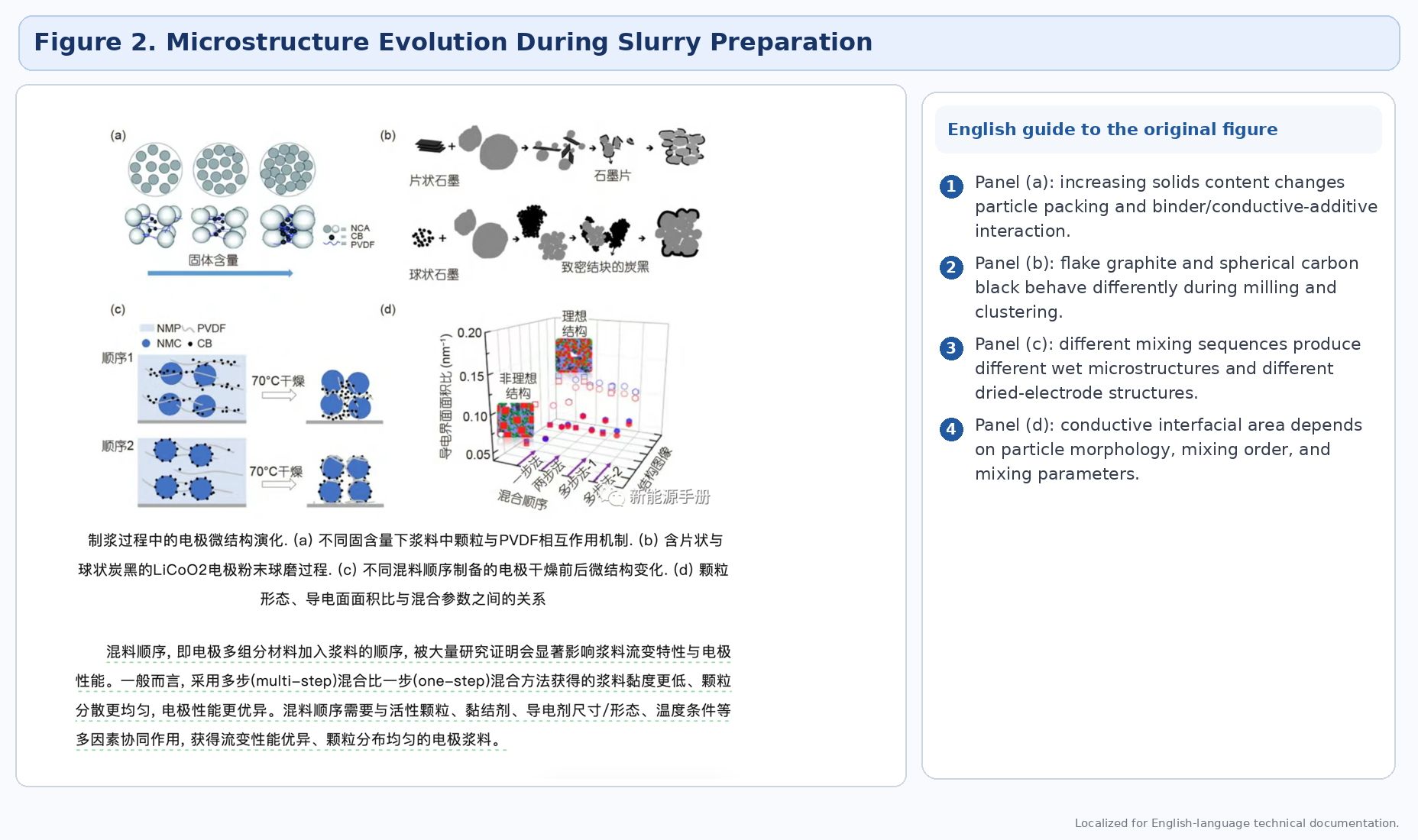
3. Main slurry-preparation routes
The source article identifies several preparation routes used for cathode slurries: wet process, semi-dry process, dry process, solvent-wetting process, and twin-screw continuous preparation. Each route changes how solvent, binder, conductive additive, and active material interact, and therefore changes wetting efficiency, cluster breakup, binder adsorption, and rheological evolution.
3.1 Wet process
In the wet process, binder solution is prepared first and then conductive additive and active material are introduced in stages. This route is conventional and well understood, but it can require longer preparation time and more liquid-handling steps.
3.2 Semi-dry process
The semi-dry route introduces the binder solution into a high-solids kneading stage, then dilutes and disperses to the final process window. It can improve wetting efficiency in some dense systems, but requires tighter control of solids loading and equipment torque.
3.3 Dry process
The dry route skips the separate binder-solution preparation stage. Powders are premixed first, then wetted and dispersed in stages. It can reduce time and process complexity, but the final dispersion step must be strong enough to avoid unstable slurry structure.
3.4 Solvent-wetting process
This hybrid route partially wets the system before final binder integration. It aims to combine some advantages of dry and wet preparation while shortening the total route.
3.5 Twin-screw continuous preparation
Twin-screw preparation supports continuous feeding and a more consistent residence time. The source highlights its advantage in continuity and process consistency, while also warning about wear, cleaning difficulty, and maintenance burden.
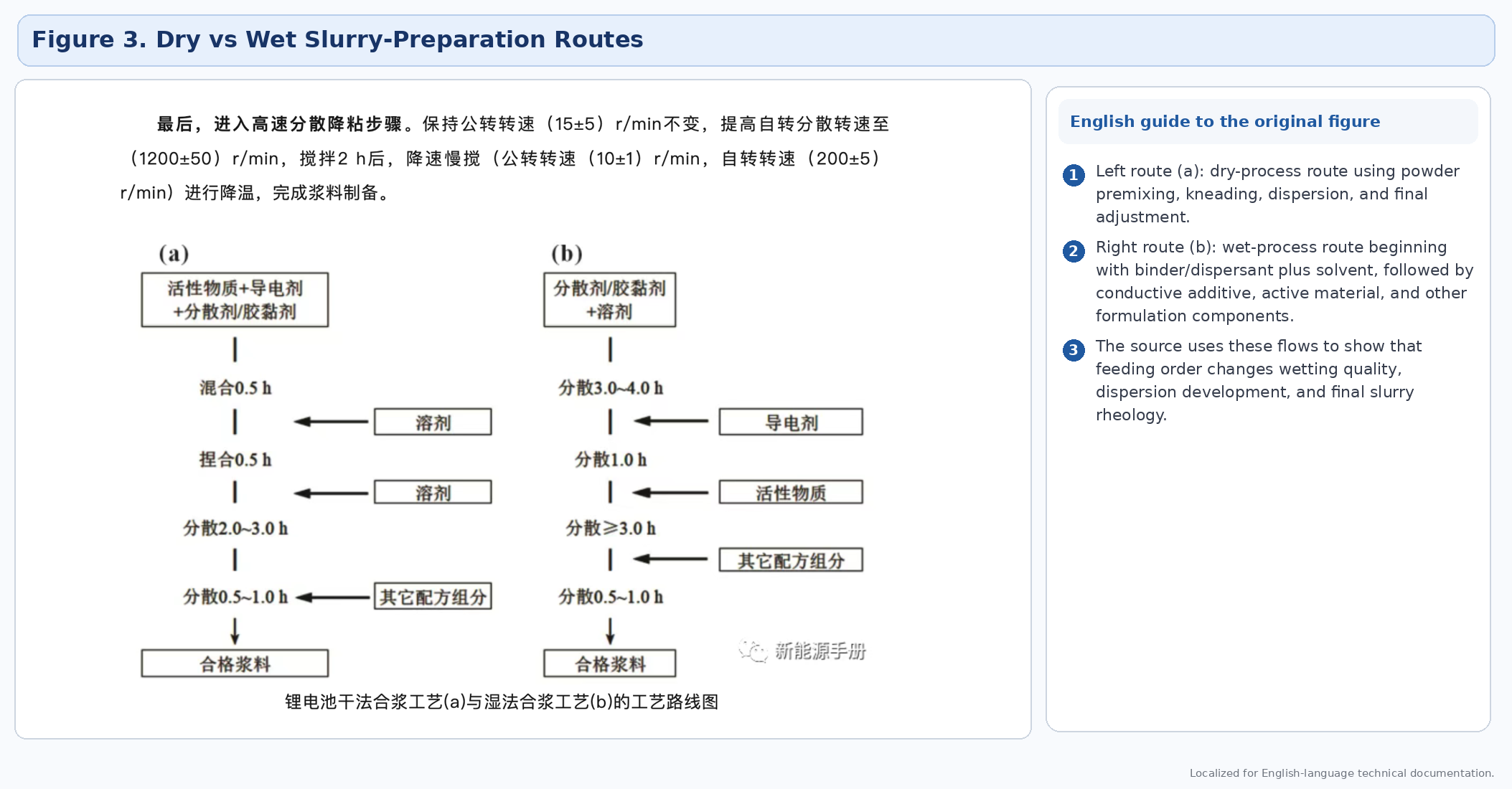
Comparison of preparation routes
| Route | Binder introduction | Process character | Typical advantage | Typical concern |
|---|---|---|---|---|
| Wet | Binder solution prepared first | Conventional and widely used | Good control of binder dissolution | Longer process time; more liquid handling |
| Semi-dry | Binder solution added into high-solids kneading stage | Dense intermediate slurry | Improved wetting efficiency in some systems | Needs careful solids and torque control |
| Dry | PVDF introduced without separate binder-solution stage | Powder-dominant early stage | Simplified route and shorter preparation | Needs strong final dispersion |
| Solvent-wetting | PVDF added later after initial wetting/dispersing | Hybrid route | Process simplification with acceptable stability | Late-stage binder integration must be controlled |
| Twin-screw | Continuous feed of paste, solvent, and active material | Continuous processing | Stable residence time and scalable output | Wear, cleaning difficulty, maintenance burden |
4. Factors affecting slurry rheology and dispersion
The article explains that slurry rheology is not controlled by one single variable. Instead, active-material properties, binder chemistry, conductive-additive morphology, solvent choice, additive package, pH, temperature, and mixing sequence all shape the final internal structure.
4.1 Active material
Particle size, surface chemistry, and particle-size distribution affect both dispersion and rheological structure. Fine particles can create stronger interaction networks and higher gel-like structure, while larger particles may produce a more fluid system.
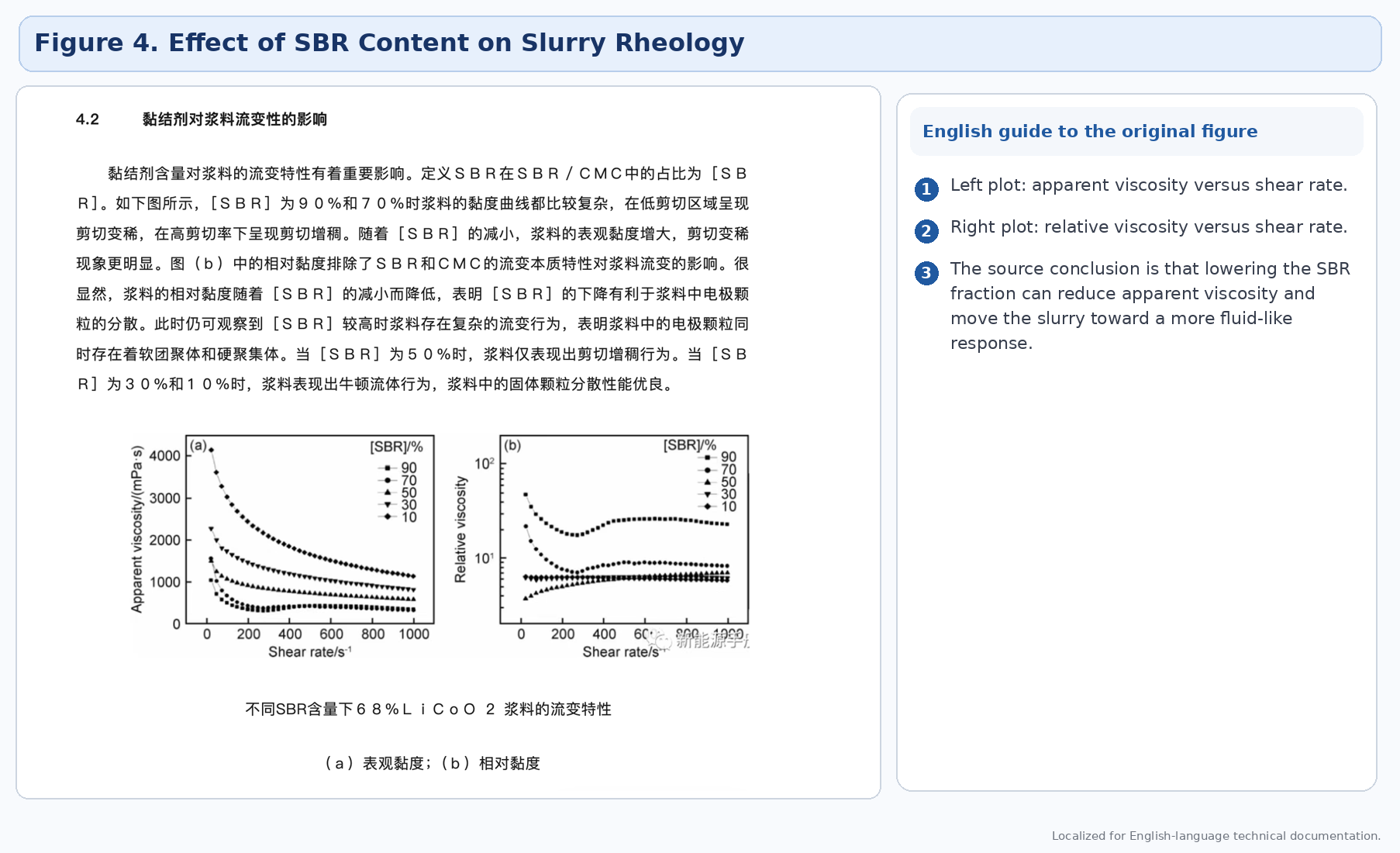
4.2 Binder
Binder type and binder ratio strongly influence slurry flow behavior, internal structure, and final electrode adhesion. The source uses SBR/CMC examples to demonstrate that changing binder composition can shift the slurry between more complex rheological behavior and more fluid-like response.
4.3 Conductive additive
Conductive additives differ greatly in morphology, surface area, and ease of dispersion. Higher-surface-area additives can reduce required dosage, but they are often more difficult to disperse and can raise slurry viscosity sharply if the process window is not well tuned.
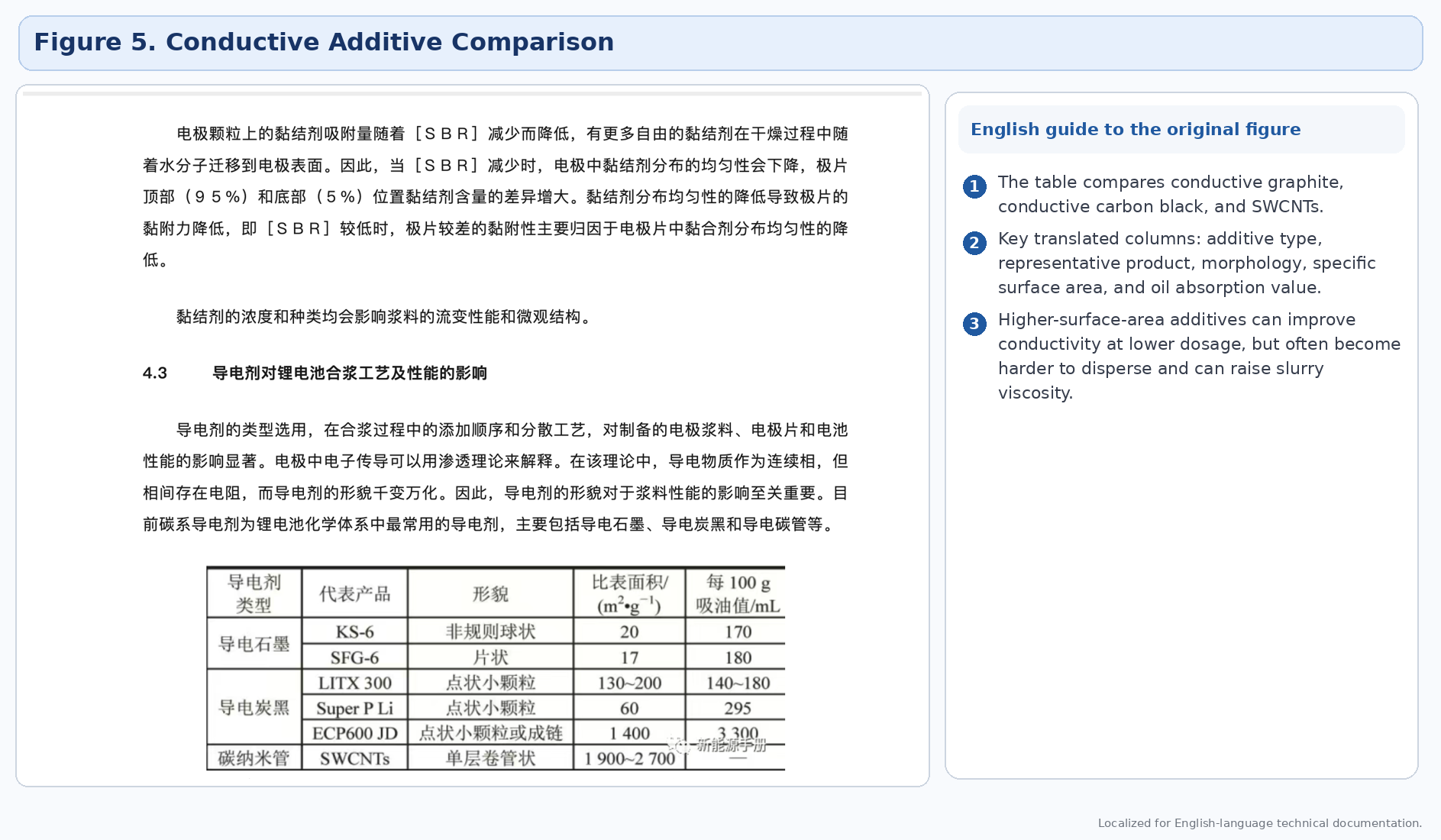
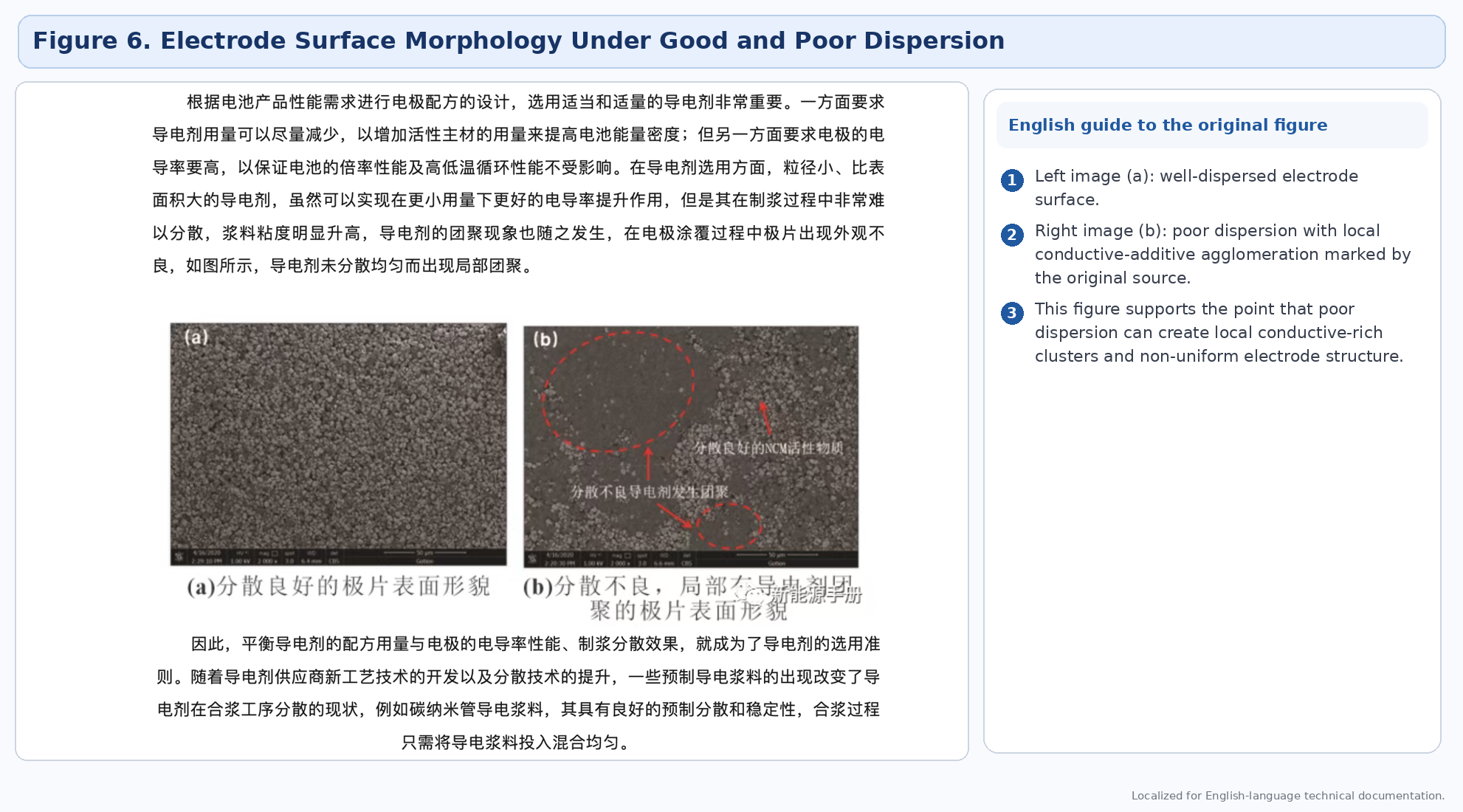
4.4 Solvent
Solvent selection affects binder affinity, particle wetting, viscosity, and redistribution during drying. The source contrasts organic and water-based systems and argues that lower-viscosity organic systems can allow stronger redistribution of graphite during drying, while water-based systems may preserve composition more uniformly through thickness.
4.5 Dispersant additives
Dispersant additives can alter contact angle, cluster interaction, and suspension stability. The source uses octanol as an example of a secondary fluid that can generate capillary-force effects in a CMC aqueous system, thereby modifying rheological behavior.
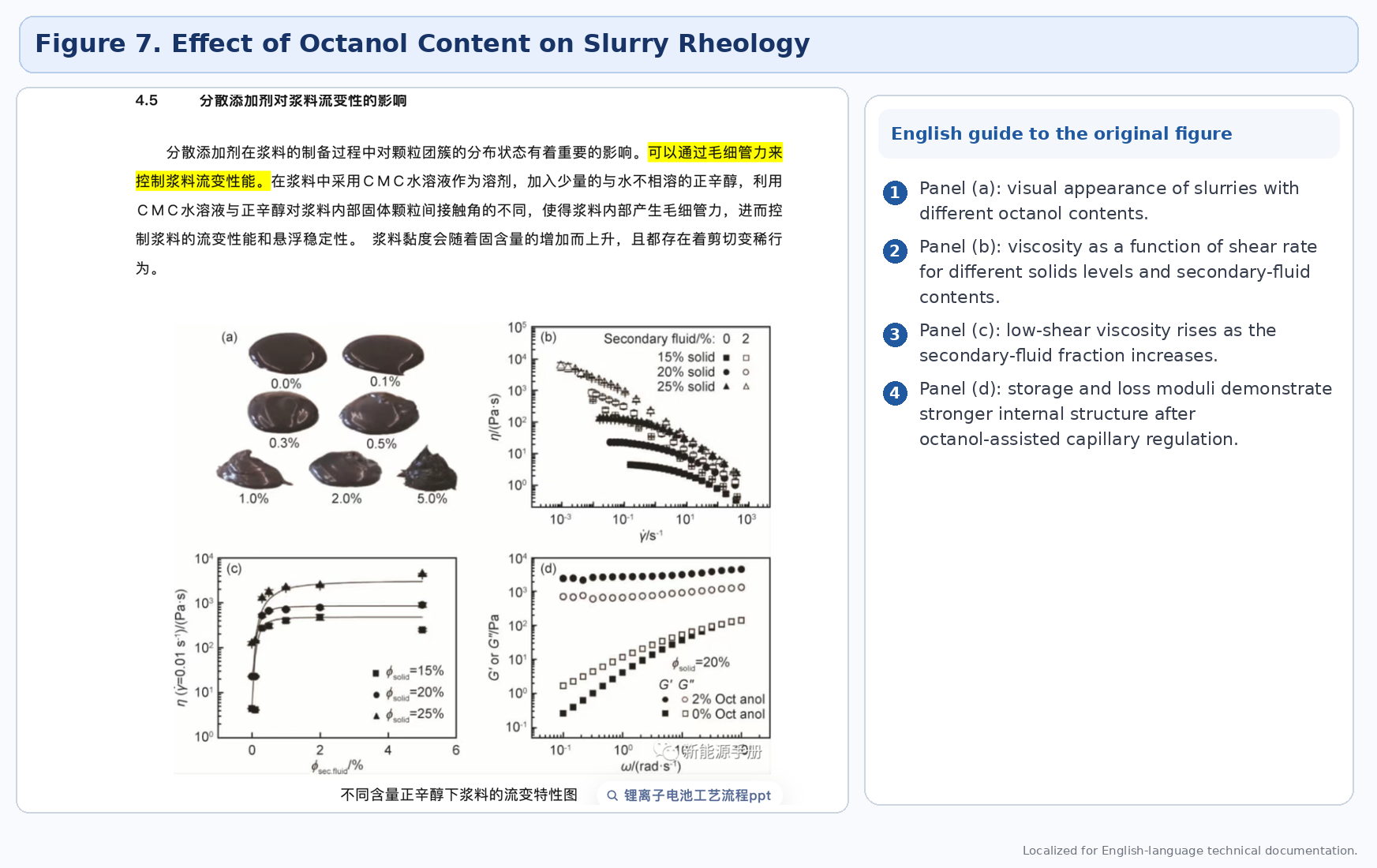
4.6 pH and temperature
pH changes the ionization state of polymer chains and therefore changes viscosity, dispersion stability, and sedimentation behavior. Temperature affects Brownian motion, viscous force balance, and adsorption behavior between fine particles and conductive carbon.
4.7 Mixing sequence
The source repeatedly emphasizes that mixing order matters. Even when exactly the same raw materials and amounts are used, one-step and multi-step mixing routes can produce different viscosity, different particle distribution, and different electrochemical performance.
5. Slurry evaluation methods
In production, the most practical methods are those that reveal both flow behavior and internal structural state.
| Indicator | What it reveals | Typical method |
|---|---|---|
| Apparent viscosity | Basic pumpability and coatability | Rotational viscometer |
| Rheological curve | Shear-thinning, yield response, non-Newtonian behavior | Rheometer |
| Viscoelasticity | Balance of liquid-like and solid-like behavior | Oscillatory rheology |
| Thixotropy | Structural breakdown and rebuild under shear history | Hysteresis loop test |
| Solids content | Concentration stability and drying load | Drying / mass-loss method |
| Particle fineness | Agglomeration level and uniformity | Grind gauge / fineness test |
| ERT or related imaging | Conductive-particle distribution state | Electrical tomography / imaging-based methods |
6. ERT as a tool for conductive-particle dispersion analysis
The article discusses electrical resistance tomography as a way to visualize conductivity distribution inside cathode slurry before drying. In LiCoO₂ cathode slurry, conductive carbon black must distribute uniformly around active particles to establish stable electronic pathways. ERT can reveal whether carbon-black agglomeration still exists and how dispersion evolves with mixing time.
7. Practical strategies to improve slurry performance
In water-based systems, dispersants or surfactants are often needed because hydrogen bonding and electrostatic interactions can make agglomeration more severe. In oil-based systems, weak polymer-network formation can be helpful for anti-settling performance. Multi-step powder addition often provides more uniform dispersion than one-time addition.
8. Why these lessons matter for SWCNT conductive slurries
Although the article is not limited to carbon nanotubes, its conclusions are highly relevant for SWCNT conductive slurries. SWCNT systems can build long-range conductive pathways at very low dosage, but that advantage depends critically on dispersion quality, solvent fit, binder chemistry, and mixing discipline.
9. Conclusion
This revised document upgrades the earlier English version by replacing raw source-image insertions with English-localized figure panels. The result is more suitable for English-language technical sharing, customer education, and website publication.
Related Technical Pages
Discuss slurry stability, rheology, and conductive-network fit with our team.
If you are comparing CNT or SWCNT slurry routes, we can help define first-pass checks around rheology, dispersion quality, hold-time stability, and electrode-performance validation.