Carbon Nanotubes as Conductive Additives for Lithium-Ion Batteries: Progress in Industrialization and Application
A long-form engineering article adapted from a technical review paper, preserving the original performance tables, application comparisons, and industrialization logic for CNT conductive additives across lithium-ion battery systems.
Source note. This article is adapted for website reading from the PDF review Carbon Nanotubes as Conductive Additives for Lithium-Ion Batteries: Progress in Industrialization and Application (DOI: 10.19799/j.cnki.2095-4239.2025.0864). The technical framing, tables, and quantitative comparisons are preserved as closely as possible in HTML form so engineers can review the material without relying on a PDF download.
Abstract
Carbon nanotubes (CNTs), built from sp²-hybridized carbon atoms, are one-dimensional nanomaterials with high intrinsic conductivity and strong mechanical properties. Through a line-contact and line-bridging mechanism, CNTs can build efficient three-dimensional conductive networks in lithium-ion battery electrodes, improving electron transport, enabling lower additive loading, and supporting difficult systems such as high-nickel cathodes, silicon-based anodes, thick electrodes, and some solid-state architectures.
The source review argues that multi-walled CNTs (MWCNTs) combined with carbon black have already achieved broad industrial use, while single-walled CNTs (SWCNTs) still face challenges in bulk manufacturing, dispersion, and cost. At the same time, the review shows why CNTs are increasingly evaluated as structural conductive-network materials rather than as simple conductivity additives.
Introduction
As lithium-ion batteries move toward higher energy density, longer cycle life, and faster charging, conductive additives become more important in electrode design. Conventional carbon black systems rely on point-contact conduction and often require 2-5 wt% loading to achieve a stable conductive network. That approach reduces the proportion of active material in the electrode and becomes more limiting in thick electrodes, high-loading systems, and chemistries with large volume change.
CNTs change the problem because their high aspect ratio enables long-range conductive pathways at lower dosage. The review positions this as one of the main reasons CNTs are increasingly important in high-nickel ternary systems, silicon-containing anodes, and all-solid-state batteries.
Overview of CNTs as conductive additives
CNTs are generally classified into single-walled CNTs (SWCNTs) and multi-walled CNTs (MWCNTs). As conductive additives, their value comes from their very high aspect ratio, strong intrinsic conductivity, and ability to form a connected network with lower addition levels than conventional carbon black.
Table 1. Comparison of intrinsic performance parameters
| Performance Parameter | SWCNTs | MWCNTs | Conductive Carbon Black (e.g. N990) |
|---|---|---|---|
| Diameter (nm) | 0.7-3.0 | 1.3-100 | 20-50 (primary particle size) |
| Electrical conductivity (S/cm) | 10^6-10^8 | 10^5 | 10^2 |
| Thermal conductivity [W/(m·K)] | 3000-6000 | 2000-3000 | 0.1-0.5 |
| Specific surface area (m²/g) | 400-1300 | 200-400 | 50-1500 |
| Tensile strength (GPa) | 120 | 10-60 | Discontinuous phase |
| Addition mass fraction (wt%) | 0.2-1.0 | 0.5-1.5 | 2-5 |
| Dispersibility | Extremely difficult | Good | Excellent |
| Conductive mechanism | 1D (Excellent) | Mixed 1D/3D (Good) | Point contact (Average) |
The source review highlights the conductivity gap clearly: SWCNTs are reported at 10^6-10^8 S/cm, MWCNTs around 10^5 S/cm, and conductive carbon black around 10^2 S/cm. Just as importantly, the review places typical addition ranges at roughly 0.2-1.0 wt% for SWCNTs, 0.5-1.5 wt% for MWCNTs, and 2-5 wt% for traditional carbon black systems.
Mechanisms of action in lithium-ion batteries
Three-dimensional conductive network construction
The review centers on the idea that CNTs establish three-dimensional conductive networks through line-contact and line-bridging effects. This can reduce interfacial resistance, improve electron transport, and preserve a higher proportion of active material by lowering conductive-additive dosage.
Interface stability regulation and CEI/SEI modification
Surface-functionalized CNTs such as CNT-COOH can influence electrolyte decomposition products and help guide denser, more stable CEI or SEI formation. On cathodes, the review highlights suppression of electrolyte decomposition and transition-metal dissolution. On anodes, doped CNTs are described as helping regulate surface electron distribution and support more uniform SEI development.
Mechanical enhancement and volume-change buffering
In silicon-based systems, CNTs can act as a flexible conductive scaffold around active particles. The review points to volume expansion on the order of 300% in silicon systems and describes CNTs as helping absorb and redistribute that stress, reducing fracture and preserving conductive continuity over repeated cycling.
Industrialization and scale-up manufacturing
The source review divides CNT industrialization into several phases: early technical exploration before 2014, scale-up and domestic substitution from 2014 to 2021, and rapid expansion with stronger competition from 2022 onward. It notes strong downstream demand growth and cites 2024 China power-battery installed capacity of 548.4 GWh, up 41.5% year over year, as a key demand driver for upstream conductive-additive materials.
The review also describes three broad industry characteristics: strong demand growth, high industry concentration, and rapid technology iteration. MWCNTs are already widely used in industrial composite conductive systems, while SWCNTs remain more challenging because of cost, bulk manufacturing, and dispersion complexity.
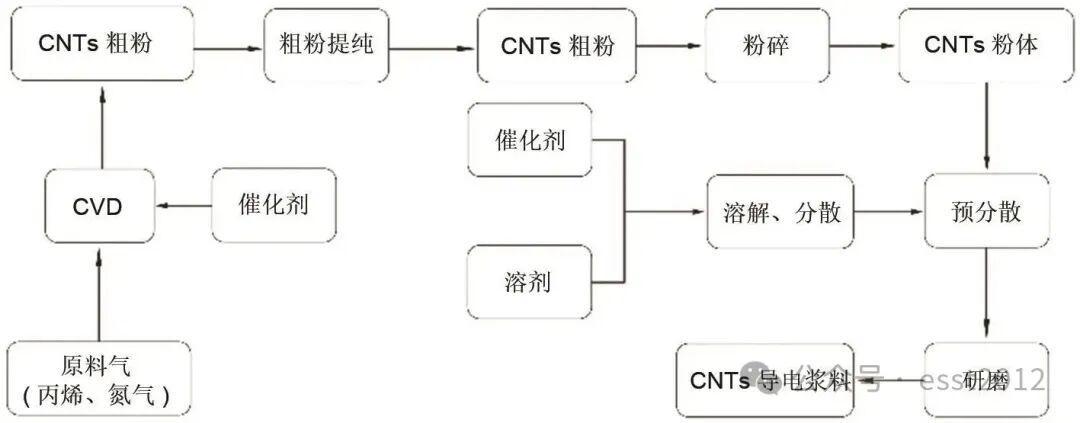
Bulk production technology
Chemical vapor deposition (CVD) is identified as the mainstream industrial production route. Fluidized-bed CVD is emphasized as the major path for large-scale MWCNT production because it supports continuous operation and favorable heat and mass transfer. The review also discusses catalyst evolution from single-metal systems to multi-element alloy systems, along with AI-assisted process control for tighter diameter, wall-number, and defect-density control.
Post-processing matters as much as synthesis
The review treats purification, dispersibility control, surface modification, and drying as key engineering steps. Acid oxidation, high-temperature annealing, sand milling, ultrasonic treatment, surfactant/polymer support, silane chemistry, element doping, and spray-drying control are all described as part of the real performance pathway. This is highly relevant for industrial screening because CNT value disappears quickly when dispersion and redispersibility are poorly controlled.

Application fields and product forms
The review states that CNT conductive-additive loading is typically around 0.5-1.5 wt%, significantly lower than the 2.0-5.0 wt% range of traditional conductive carbon black. It describes this lower dosage as helpful for reducing internal resistance, improving energy density, and supporting systems with large volume change. It also notes that conductive pastes remain the primary CNT product form today, typically using NMP or deionized water as the solvent system at about 3-6 wt% concentration.
That observation aligns with what many engineering teams see in practice: slurry or paste format often reduces the burden of in-house CNT dispersion during initial evaluation, while dry-electrode development may increase future demand for powder-form CNT conductive additives.
Table 2. Comparison of application performance between CNTs and traditional carbon-based conductive agents
| Performance Parameter | Ternary Cathode CNTs | Ternary Traditional Carbon | Silicon-Anode CNTs | Silicon-Anode Traditional Carbon | LFP CNTs | LFP Traditional Carbon | ASSB SWCNTs | ASSB Special Agent |
|---|---|---|---|---|---|---|---|---|
| Addition mass fraction (%) | 0.5-1.0 | 2.0-3.0 | 1.0-1.5 | 3.0-5.0 | 0.8-1.2 | 2.0-3.0 | 0.2-0.5 | 1.5-2.5 |
| Internal resistance (Ω) | 8-10 | 12-15 | 10-12 | 18-22 | 5-7 | 8-10 | 8-10 | 15-18 |
| Rate performance (%) | 105-108 | 82-85 | 105-110 | 75-78 | 102-105 | 85-88 | 115-120 | 78-82 |
| Cycle life (cycles) | 1350-1450 | 800-1000 | 600-700 | 300-400 | 3500-4000 | 2500-3000 | 250-350 | 100-200 |
| Energy density (Wh/kg) | 265-275 | 240-250 | 450-480 | 380-400 | 165-170 | 155-160 | 340-360 | 280-300 |
System compatibility assessment
Cathode systems
The review covers LFP, high-nickel ternary cathodes, and high-voltage emerging cathodes. In LFP, the main issue is low intrinsic conductivity and stronger polarization in thick-electrode and fast-charging scenarios. In high-nickel ternary systems such as NCM811, the review highlights low intrinsic conductivity, structural instability, and transition-metal dissolution under high-voltage cycling. In high-voltage LiCoO₂ and lithium-rich layered oxides, it emphasizes electrolyte decomposition and unstable CEI growth above 4.5 V.
Across these systems, CNTs are positioned as helping through conductive-network construction, interface regulation, and mechanical integrity support. The review specifically calls out CNT-COOH in NCM811, along with thermal-management and interfacial-regulation advantages for SWCNTs in high-voltage systems.
Table 4. Performance comparison in different cathode systems
| Cathode Type | CNT Type | Addition Mass Fraction (%) | Key Performance Improvement |
|---|---|---|---|
| LFP | MWCNTs/CB composite | 0.8-1.5 | 0.5 C capacity: 146.32 mAh/g; 91.78% retention after 2165 cycles |
| NCM | CNT-COOH/CB composite | 0.5-1.2 | 73.8% retention after 200 cycles at 1 C; 53.1% retention at 10 C |
| LMO | (8,0)-type SWCNTs | 0.3-0.8 | 5 C discharge-capacity retention increased to 82% and Mn dissolution inhibited |
| NMCA | COF/CNTs | 1.0-2.0 | Specific capacity: 314 mAh/g; 88% retention after 10,000 cycles |
| LRLO | N-S-CNTs | 1.0-2.5 | Improved electrical conductivity and charge-discharge performance |
| P0.02-nrNCM | Bulk doping synergistic with CNTs | — | 1 C capacity: 166.07 mAh/g; 92.4% retention after 100 cycles |
Anode systems
For anodes, the review focuses on poor conductivity, major volume expansion, and unstable SEI formation in silicon-based materials. It distinguishes ultra-high-silicon and pure-silicon systems, where SWCNTs are described as ideal for flexible network building, from medium- to high-silicon systems where MWCNTs are often preferred for cost and process compatibility.
The review cites several silicon-system outcomes, including reduced electron-transport resistance, lower electrode expansion, better coulombic efficiency, and commercial pilot-scale results in NCM811//SiOx/C batteries reported at 450-480 Wh/kg and 600-700 cycles with at least 80% capacity retention under 1 C cycling.
Table 5. Silicon-anode comparison
| Performance Index | Super P | MWCNTs | SWCNTs | Core Action Mechanism |
|---|---|---|---|---|
| First discharge specific capacity (mAh/g) | 1200-1500 | 1500-1800 | 1800-2200 | 3D CNT networks enhance electronic conduction |
| Capacity retention after 100 cycles (%) | 55-65 | 60-70 | 70-80 | CNT networks buffer volume expansion |
| Average coulombic efficiency (%) | 97.0-98.0 | 99.0-99.3 | 99.5-99.8 | CNTs facilitate stable SEI formation and reduce irreversible lithium consumption |
| Electrode expansion rate (%) | 300-400 | 150-200 | 100-150 | CNTs inhibit overall expansion |
| Interfacial impedance (Ω) | 80-100 | 40-50 | 30-40 | CNTs enhance interfacial coupling and ion transport |
Next-generation battery technologies
The review also extends CNT relevance to all-solid-state batteries (ASSBs), sodium-ion batteries (SIBs), and lithium-sulfur batteries (LSBs). In ASSBs, high-purity SWCNT pastes are described as enabling interpenetrating ion/electron dual-conducting networks with sulfide electrolytes and pushing projected energy density beyond 500 Wh/kg in the cited work. In SIBs and LSBs, CNT-based conductive frameworks are presented as helping electron transport, sulfur conversion, and shuttle suppression.
Comprehensive performance comparison and selection
The paper’s selection logic is practical: high-nickel ternary and silicon-based anodes need CNTs that combine conductive continuity, mechanical flexibility, and interface regulation; low-cost systems like LFP prioritize MWCNT cost-effectiveness and dispersion stability; and ASSBs demand extremely high-purity, high-aspect-ratio SWCNTs. It also emphasizes that application selection should be matched to energy-density goals, rate requirements, and cost constraints rather than to one universal CNT story.
Table 6. Compatibility of CNT conductive agents with different battery systems
| Battery System | Recommended CNT Type | Addition Mass Fraction (%) | Key Improvement Indicators | Mechanism of Action |
|---|---|---|---|---|
| LFP cathode | MWCNTs | 0.8-1.2 | Energy density 165-170 Wh/kg; 3C retention >102% | Optimization of thick-electrode conductive network |
| High-Ni ternary | MWCNTs-COOH | 0.5-1.0 | Cycle life >1350 cycles; internal resistance 8-10 Ω | CEI stabilization and suppression of transition-metal dissolution |
| Silicon-based anode | N-doped SWCNTs | 1.0-1.5 | Capacity retention >600 cycles; expansion rate <83% | Net-pocket structure and Si-C bonding |
| ASSBs | SWCNTs | 0.2-0.5 | Energy density 340-360 Wh/kg; interfacial impedance <35 Ω | Dual ion/electron pathways |
| LSB | Ni3V2O8@CNTs | 1.5-2.0 | Decay rate 0.0334% over 1500 cycles | Polysulfide adsorption and catalysis |
| SIBs | MWCNTs | 1.0-1.5 | Specific capacity >118 mAh/g; cycles >500 | Accelerated electron transport |
Conclusions and perspectives
The review argues that CNT conductive additives are moving from the category of auxiliary conductivity materials toward multifunctional core materials. The reason is not only conductivity. It is the combination of network construction, interfacial stabilization, and mechanical buffering in chemistries that are already pushing conventional carbon-additive logic to its limit.
It also makes the current bottlenecks clear: SWCNT scale-up remains expensive, bulk production still faces technical barriers, dispersion complexity constrains industrial adoption, and greener, lower-cost manufacturing routes are still needed. The future directions it highlights are precision catalyst and process design, greener and more intelligent manufacturing, and broader use in sodium-ion, solid-state, sulfur, and flexible battery systems.
For engineering teams, the practical lesson is straightforward: CNT selection should be treated as a system-design and process-fit question. The right answer depends on chemistry, loading strategy, dispersion route, manufacturing compatibility, and the performance bottleneck that the electrode is actually trying to solve.
Related Technical Pages
- SWCNT engineering guide for conductive slurry evaluation
- SWCNT vs carbon black in thick electrodes
- SWCNT slurry vs powder for process-route decisions
- High-Ni cathode fit for conductive-network screening
- Silicon-anode fit for conductive-network evaluation
- LFP and ESS applications for CNT conductive systems
Use CNT selection as a system-fit decision, not only an additive-choice decision.
If your team is comparing SWCNT powder, conductive slurry, or hybrid conductive systems across high-Ni, silicon, LFP, or solid-state-adjacent programs, we can help narrow the first technical questions.